Compton Scattering
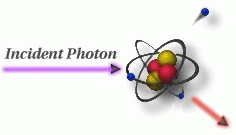 As mentioned on the previous page, Compton scattering occurs when the incident x-ray photon is deflected from its original path by an interaction with an electron. The electron is ejected from its orbital position and the x-ray photon loses energy because of the interaction but continues to travel through the material along an altered path. Energy and momentum are conserved in this process. The energy shift depends on the angle of scattering and not on the nature of the scattering medium. Since the scattered x-ray photon has less energy, it has a longer wavelength and less penetrating than the incident photon.
As mentioned on the previous page, Compton scattering occurs when the incident x-ray photon is deflected from its original path by an interaction with an electron. The electron is ejected from its orbital position and the x-ray photon loses energy because of the interaction but continues to travel through the material along an altered path. Energy and momentum are conserved in this process. The energy shift depends on the angle of scattering and not on the nature of the scattering medium. Since the scattered x-ray photon has less energy, it has a longer wavelength and less penetrating than the incident photon.
Compton effect was first observed by Arthur Compton in 1923 and this discovery led to his award of the 1927 Nobel Prize in Physics. The discovery is important because it demonstrates that light cannot be explained purely as a wave phenomenon. Compton's work convinced the scientific community that light can behave as a stream of particles (photons) whose energy is proportional to the frequency.
The change in wavelength of the scattered photon is given by:
|
Where:
|
= | wavelength of incident x-ray photon | |
| = | wavelength of scattered x-ray photon | ||
| h | = | Planck's Constant: The fundamental constant equal to the ratio of the energy E of a quantum of energy to its frequency ν: E=hν. | |
| me | = | the mass of an electron at rest | |
| c | = | the speed of light | |
| = | The scattering angle of the scattered photon |
The applet below demonstrates Compton scattering as calculated with the Klein-Nishina formula, which provides an accurate prediction of the angular distribution of x-rays and gamma-rays that are incident upon a single electron. Before this formula was derived, the electron cross section had been classically derived by the British physicist and discoverer of the electron, J.J. Thomson. However, scattering experiments showed significant deviations from the results predicted by Thomson's model. The Klein-Nishina formula incorporates the Breit-Dirac recoil factor, R, also known as radiation pressure. The formula also corrects for relativistic quantum mechanics and takes into account the interaction of the spin and magnetic moment of the electron with electromagnetic radiation. Quantum mechanics is
a system of mechanics based on quantum theory to provide a consistent explanation of both electromagnetic wave and atomic structure.
The applet shows that when a photon of a given energy hits an atom, it is sometimes reflected in a different direction. At the same time, it loses energy to an electron that is ejected from the atom. Theta is the angle between the scattered photon direction and the path of the incident photon. Phi is the angle between the scattered electron direction and the path of the incident photon.
Set the energy (hv) to zero (default value) to get the Thomson scattering curve. The incident photon has an energy of hv, which can be set in the applet above the interactive graphic. To change this, and any other number in the applet, simply click in the box and retype the number. Don't forget to press the "enter" key on the keyboard to make the new value effective. Phi represents the angle the resultant photon makes with the X-axis. This can also be changed by simply clicking on the white area. Theta is the angle the resultant electron makes with the X-axis. Whenever a new angle is inputted, a red dot should appear on the graph to the right. This represents the intensity ratio for a given degree, as predicted by the Klein-Nishina formula. If the ratio is high, this means that the intensity of the resulting photon is close to that of the incident photon. Theta is the angle between the scattered photon direction and the path of the incident photon. Phi is the angle between the scattered electron direction and the path of the incident photon.