Solid State Structure
In the previous pages, some of the mechanisms that bond together the multitude of individual atoms or molecules of a solid material were discussed. These forces may be primary chemical bonds, as in metals and ionic solids, or they may be secondary van der Waals’ forces of solids, such as in ice, paraffin wax and most polymers. In solids, the way the atoms or molecules arrange themselves contributes to the appearance and the properties of the materials.
Atoms can be gathered together as an aggregate through a number of different processes, including condensation, pressurization, chemical reaction, electrodeposition, and melting. The process usually determines, at least initially, whether the collection of atoms will take to form of a gas, liquid or solid. The state usually changes as its temperature or pressure is changed. Melting is the process most often used to form an aggregate of atoms. When the temperature of a melt is lowered to a certain point, the liquid will form either a crystalline solid or and amorphous solid.
Amorphous Solids
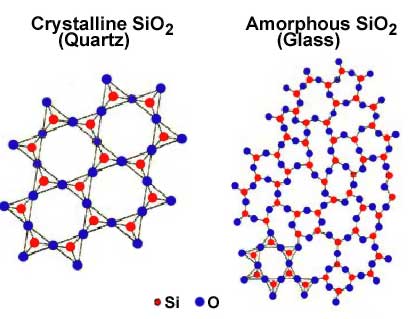
A solid substance with its atoms held apart at equilibrium spacing, but with no long-range periodicity in atom location in its structure is an amorphous solid. Examples of amorphous solids are glass and some types of plastic. They are sometimes described as supercooled liquids because their molecules are arranged in a random manner some what as in the liquid state. For example, glass is commonly made from silicon dioxide or quartz sand, which has a crystalline structure. When the sand is melted and the liquid is cooled rapidly enough to avoid crystallization, an amorphous solid called a glass is formed. Amorphous solids do not show a sharp phase change from solid to liquid at a definite melting point, but rather soften gradually when they are heated. The physical properties of amorphous solids are identical in all directions along any axis so they are said to have isotropic properties, which will be discussed in more detail later
.
Crystalline Solids
More than 90% of naturally occurring and artificially prepared solids are crystalline. Minerals, sand, clay, limestone, metals, carbon (diamond and graphite), salts ( NaCl, KCl etc.), all have crystalline structures. A crystal is a regular, repeating arrangement of atoms or molecules. The majority of solids, including all metals, adopt a crystalline arrangement because the amount of stabilization achieved by anchoring interactions between neighboring particles is at its greatest when the particles adopt regular (rather than random) arrangements. In the crystalline arrangement, the particles pack efficiently together to minimize the total intermolecular energy.
The regular repeating pattern that the atoms arrange in is called the crystalline lattice. The scanning tunneling microscope (STM) makes it possible to image the electron cloud associated individual atoms at the surface of a material. Below is an STM image of a platinum surface showing the regular alignment of atoms.
 Courtesy: IBM Research, Almaden Research Center.
Courtesy: IBM Research, Almaden Research Center.
Crystal Structure
Crystal structures may be conveniently specified by describing the arrangement within the solid of a small representative group of atoms or molecules, called the ‘unit cell.’ By multiplying identical unit cells in three directions, the location of all the particles in the crystal is determined. In nature, 14 different types of crystal structures or lattices are found. The simplest crystalline unit cell to picture is the cubic, where the atoms are lined up in a square, 3D grid. The unit cell is simply a box with an atom at each corner. Simple cubic crystals are relatively rare, mostly because they tend to easily distort. However, many crystals form body-centered-cubic (bcc) or face-centered-cubic (fcc) structures, which are cubic with either an extra atom centered in the cube or centered in each face of the cube. Most metals form bcc, fcc or Hexagonal Close Packed (hpc) structures; however, the structure can change depending on temperature. These three structures will be discussed in more detail on the following page.
Crystalline structure is important because it contributes to the properties of a material. For example, it is easier for planes of atoms to slide by each other if those planes are closely packed. Therefore, lattice structures with closely packed planes allow more plastic deformation than those that are not closely packed. Additionally, cubic lattice structures allow slippage to occur more easily than non-cubic lattices. This is because their symmetry provides closely packed planes in several directions. A face-centered cubic crystal structure will exhibit more ductility (deform more readily under load before breaking) than a body-centered cubic structure. The bcc lattice, although cubic, is not closely packed and forms strong metals. Alpha-iron and tungsten have the bcc form. The fcc lattice is both cubic and closely packed and forms more ductile materials. Gamma-iron, silver, gold, and lead have fcc structures. Finally, HCP lattices are closely packed, but not cubic. HCP metals like cobalt and zinc are not as ductile as the fcc metals.