Electric Charge and Fields
After reading this section you will be able to do the following:
- Explain the electrical differences between electrons and protons.
- Predict what happens when electric charges interact with other charges.
Electrons
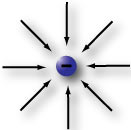
Electrons, like the one shown on the left, are the smallest and lightest of the particles in an atom. Electrons are in constant motion as they circle around the nucleus of that atom. Electrons are said to have a negative charge with a value of -1.602x10^-19 coulombs (ku`-lums). Coulombs (C) is the unit used to describe how much charge is present. It is the charge carried by 6.25 x 10^18 electrons. 6.25 x 10^18 is scientific notation for 6,250,000,000,000,000,000.
Protons
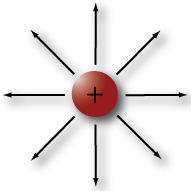
Protons, like the one shown on the right, are much larger and heavier than electrons and reside in the atom's nucleus. Protons have a positive electrical charge with a value of +1.602x10^-19 coulombs. A good way to remember what charge protons have is to remember both proton and positive charge start with "P."
Electrostatic Fields
Non-zero charges are surrounded by a kind of invisible force field called an electrostatic field. An electrostatic field comes out of a positive charge, like is shown by the outward arrows in the proton diagram above, and goes into a negative charge (see the electron diagram). In other words, the electrostatic fields for positive charges and negative charges have opposite polarity. Notice the negative electron and the positive proton have the same number of force field lines in each of the diagrams. This means that the strengths of these two fields are equal and that the proton is exactly as positive as the electron is negative.
Charge Attraction
Like charges repel while unlike charges attract. Two electrons will tend to repel each other because both have a negative electrical charge. Two protons will also tend to repel each other because they both have a positive charge. On the other hand, electrons and protons will be attracted to each other because of their unlike charges.
Since the electron is much smaller and lighter than a proton, when they are attracted to each other due to their unlike charges, the electron usually does most of the moving. This is because the protons have more mass and are harder to get moving. Although electrons are very small, their negative electrical charges are still quite strong. Remember, the negative charge of an electron is the same as the positive electrical charge of a proton. This allows the atom to stay electrically balanced.
Another important fact about the electrical charges of protons and electrons is that the farther away they are from each other, the less force their electric fields have on each other. Similarly, the closer they are to each other, the more force they will experience from each other due to the electric field. The app below demonstrates what happens when a positive and negative charges are around each other.
Review
- Electrons have a negative electric charge and protons have a positive electric charge.
- Like charges repel, unlike charges attract, just like with magnets.
- Electrostatic fields surround non-zero charges.