The Valence Shell
After reading this section you will be able to do the following:
- Give an explanation of the valence shell of an atom.
- Explain what free electrons are and why they are important.
Valence Shell
The outermost shell of any atom is called the valence shell and the electrons that reside in the valence shell are called valence electrons. When the valence electrons in any atom gain sufficient energy from some outside force, they can break away from the parent atom and become what are called a free electrons. Notice that in the copper atom pictured below that the outermost shell has only one electron, which means it has one valence electron.
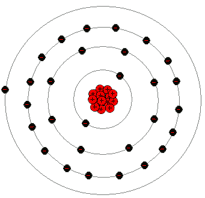
Pictured here is an atom of copper, which is much more complex than either an atom of hydrogen or helium.
Atoms with few electrons in their valence shell tend to have more free electrons since these valence electrons are more loosely bound to the nucleus. The next section discusses how free electrons behave in the presence of other atoms.
Review
- The valence shell is the outer shell of the atom.
- Some materials have a free electron in their valence shell and this electron can easily move from atom to atom.
- The free electrons are responsible for electrical current.