Isotopes
After reading this section you will be able to do the following:
- Define an isotope and explain how it is different than an element.
In our discussion of mass number we briefly mentioned isotopes. Isotopes are a form of an element that has the same number of protons but a different number of neutrons. In general, atoms want to have the same number of neutrons and protons but the number of neutrons can change. There are 3 different isotopes of hydrogen which are shown in the figure below.
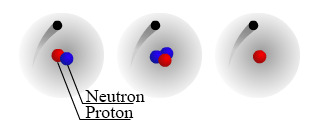
Notice that the three hydrogen atoms have the same number of protons, but a different number of neutrons. Different isotopes of an element are present in different proportions in nature. For example, light hydrogen which has no neutrons, is the most common isotope of hydrogen. The nuclear symbols of the 3 hydrogen isotopes are as follows:
H11 H12 H13
In the nuclear symbols above, the superscript represents the mass number while the subscript represents the atomic number. From these two quantities the number of neutrons present in the isotope can be calculated.
Review:
- Neutrons are all identical to each other, just as protons are.
- Atoms of a particular element must have the same number of protons but can have different numbers of neutrons.
- When an element has different variants that, while all having the same number of protons, have differing numbers of neutrons, these variants are called isotopes.