The Free Electron
After reading this section you will be able to do the following:
- Describe how elements maintain their electrical balance.
- Explain how free electrons are created and shared.
Maintaining Electrical Balance
Each basic element has a certain number protons which distinguishes each element from all other basic elements. In most elements, like the ones shown in the animation below, the number of electrons is equal to the number of protons. This maintains an electrical balance in the structure of atoms since protons and electrons have equal but opposite charges and electrostatic fields.
As previously mentioned, when the valence electrons in an atom gain sufficient energy they can break away from the parent atom and become free electrons.
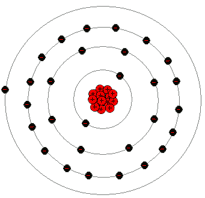
Pictured here is an atom of copper, which is much more complex than either an atom of hydrogen or helium.
Sharing Free Electrons
The copper atom, shown above, has 29 protons in its nucleus and 29 electrons orbiting its nucleus. As can be seen from the copper atom diagram, the electrons are arranged in 4 electron shells with 1 electron in the valence shell. In some materials like copper, the outer electrons are so loosely held by the atom and so close to the neighboring atoms that it is difficult to determine which electron belongs to which atom. Under these conditions, the valence or free electrons tend to drift randomly from one atom to its neighboring atoms. Under normal conditions this movement of the electrons is truly random, meaning they are moving in all directions by the same amount. The movement of free electrons between atoms can be seen in the animation below.
If some outside force acts upon the material the flow of electrons can be directed through materials and this flow is called electrical current. Materials that have free electrons and allow electrical current to flow easily are called conductors. Many materials do not have any free electrons. Because of this fact, they do not tend to share their electrons very easily and do not make good conductors of electrical currents. These materials are called insulators. There will be more information on this later.
In the next sub-unit you will learn about the role valence and free electrons play in forming molecules.
Review
- In order to maintain electrical balance the number of electrons is equal to the number of protons in most elements.
- Valence electrons can break away from the parent atom and become free electrons when they can sufficient energy.
- Free electrons can be shared among neighboring atoms.