Atomic Number and Mass Numbers
After reading this section you will be able to do the following:
- Define and determine the atomic number of an atom.
- Define and determine the mass number of an atom.
Atomic Number
The number of protons in the nucleus of an atom determines an element's atomic number. In other words, each element has a unique number that identifies how many protons are in one atom of that element. For example, all hydrogen atoms, and only hydrogen atoms, contain one proton and have an atomic number of 1. All carbon atoms, and only carbon atoms, contain six protons and have an atomic number of 6. Oxygen atoms contain 8 protons and have an atomic number of 8. The atomic number of an element never changes, meaning that the number of protons in the nucleus of every atom in an element is always the same.
Isotopes are forms of elements that have the same number of protons and therefore the same atomic number, but a different number of neutrons which affects their mass number.
Mass Number
All atoms have a mass number which is derived as follows:
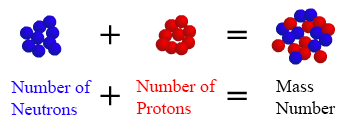
In words, the mass number is the number of neutrons in an atom of a specific element plus the number of protons in an atom of that element. For example the mass number of a regular carbon atom is 12, since a carbon atom has 6 protons and 6 neutrons in its nuclus. The mass number is approximately equal to the atomic mass, which is the mass of a single atom of a element measured in atomic mass units (amu). The atomic mass of Hydrogen is 1.00797 amu and the atomic mass of Carbon is 12.011 amu. The atomic mass is typically listed in the periodic table below the element's name.
Since isotopes have a different number of neutrons, their mass numbers and atomic masses differ from those listed in the periodic table. Isotopes are discussed more in depth later in this module.
Review:
- An element's or isotope's atomic number tells how many protons are in its atoms.
- An element's or isotope's mass number tells how many protons and neutrons in its atoms.