Interaction of Electromagnetic Radiation and Matter
It is well known that all matter is comprised of atoms. But subatomically, matter is made up of mostly empty space. For example, consider the hydrogen atom with its one proton, one neutron, and one electron. The diameter of a single proton has been measured to be about 10-15 meters. The diameter of a single hydrogen atom has been determined to be 10-10 meters, therefore the ratio of the size of a hydrogen atom to the size of the proton is 100,000:1. Consider this in terms of something more easily pictured in your mind. If the nucleus of the atom could be enlarged to the size of a softball (about 10 cm), its electron would be approximately 10 kilometers away. Therefore, when electromagnetic waves pass through a material, they are primarily moving through free space, but may have a chance encounter with the nucleus or an electron of an atom.
Because the encounters of photons with atom particles are by chance, a given photon has a finite probability of passing completely through the medium it is traversing. The probability that a photon will pass completely through a medium depends on numerous factors including the photon’s energy and the medium’s composition and thickness. The more densely packed a medium’s atoms, the more likely the photon will encounter an atomic particle. In other words, the more subatomic particles in a material (higher Z number), the greater the likelihood that interactions will occur Similarly, the more material a photon must cross through, the more likely the chance of an encounter.
When a photon does encounter an atomic particle, it transfers energy to the particle. The energy may be reemitted back the way it came (reflected), scattered in a different direction or transmitted forward into the material. Let us first consider the interaction of visible light. Reflection and transmission of light waves occur because the light waves transfer energy to the electrons of the material and cause them to vibrate. If the material is transparent, then the vibrations of the electrons are passed on to neighboring atoms through the bulk of the material and reemitted on the opposite side of the object. If the material is opaque, then the vibrations of the electrons are not passed from atom to atom through the bulk of the material, but rather the electrons vibrate for short periods of time and then reemit the energy as a reflected light wave. The light may be reemitted from the surface of the material at a different wavelength, thus changing its color.
X-Rays and Gamma Rays
X-rays and gamma rays also transfer their energy to matter though chance encounters with electrons and atomic nuclei. However, X-rays and gamma rays have enough energy to do more than just make the electrons vibrate. When these high energy rays encounter an atom, the result is an ejection of energetic electrons from the atom or the excitation of electrons. The term "excitation" is used to describe an interaction where electrons acquire energy from a passing charged particle but are not removed completely from their atom. Excited electrons may subsequently emit energy in the form of x-rays during the process of returning to a lower energy state.
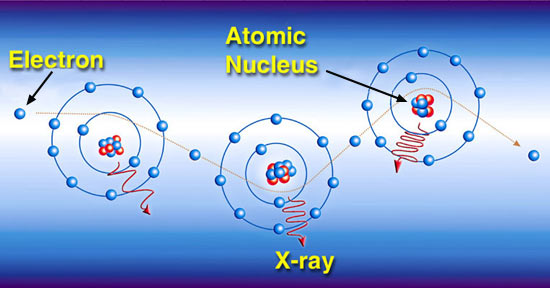
Each of the excited or liberated electrons goes on to transfer its energy to matter through thousands of events involving interactions between charged particles. With each interaction, the energy may be directed in a different direction. The higher the energy of a photon, the more likely the energy will continue traveling in the same direction. As the radiation moves from point to point in matter, it loses its energy through various interactions with the atoms it encounters. If the radiation has enough energy, it may eventually make it through the material.
Photon Interaction with Matter is Key
From the previous paragraph, it can be deduced that the energy of X- and Gamma ray photons is largely responsible for their penetrating power. Einstein linked the energy of a photon to its frequency and wavelength when he postulated that each photon carries an energy of the frequency of the wave times Planck’s constant (). The frequency of an EM wave equals the speed of light divided by the wavelength ( ). However, it should be understood that the wavelength or frequency of electromagnetic radiation does not in itself makes the EM wave more or less penetrating. The key is its interaction with matter, or more specifically, whether the photon's energy is right to excite some transition of a charged particle. For instance, microwaves penetrate glass very easily, but they are strongly absorbed by water. Move up to slightly higher frequency, and infrared is strongly absorbed by both glass and water, but both substances transmit visible light. Ultraviolet is stopped by glass, but not so readily by water.